
Shape shifting crystals put to work

June 2024
In his latest publication, Kenny describes a simple and green covalent biofunctionalization method to “plant a forest” of amphiphilic tripeptides on the surface of graphene and create stable dispersion through an aqueous enzymatic oxidation approach that is facilitated by a tyrosine phenol side chain. Kenny was able to formulate a peptide design rule necessary for dispersing graphene. Aside from tyrosine in which is necessary for covalent attachment after enzymatic oxidation, a second aromatic/aliphatic side chain group is required to enhance functionalization through non-covalent support via hydrophobic interactions with the graphene surface. Furthermore, the addition of charged moieties from terminal groups or ionizable amino acids stimulates profound interaction with water, which leads to an exceptionally stable graphene dispersion in aqueous solutions for months.

November 2023
Research in biological condensed phases has exploded over the past decade. A framework of understanding relating to how sequence dictates phase behavior in biological systems is now emerging. However, there are still gaps in understanding at molecular level that permit rational, design of peptide sequences that display sequence-controlled phase behaviors. In our new paper we used the information available from various biological and bioinspired systems described in the literature to design simple de novo repeat sequences that form dynamic interacting peptide networks. Our designs are composed of repeats of tripeptide spacers (GSG, SGS, GLG) interspersed by a pattern of adhesive amino acids (R/H and Y). We show, using a combination of computation and experiment, that spacer editing is sufficient to direct backbone structuring and phase behavior. Unexpectedly, the droplets showed intrinsic fluorescent behavior that was dictated by the peptide sequence.
April 2023
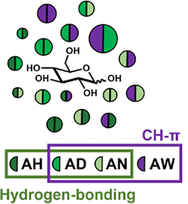
In this paper we use a systems chemistry approach to harness weak combinatorial interactions and show spontaneous emergence of network cooperativity. We study systems that are composed of selected mixtures of dipeptides chosen based on glucose binding sites from the protein data bank. A peptidase is included that can catalyze peptide bond formation, exchange and breakdown. Binding to glucose shifts the thermodynamic equilibrium toward sequences that have favorable interactions with glucose through side-chain interactions. We were able to identify networks of up to 16 interacting tetrapeptides that stabilize glucose through hydrogen bonding and CH-π interactions and highlight the crucial role of residue cooperativity in enhancing weak binding interactions, and the influence of input selection and history on the system's composition. The paper showcases fundamental systems properties that cannot be discovered or replicated with reductionist approaches.
November 2022
In this new research, Ankit and Salma designed a multi-component biomolecular condensate capable of stabilizing and confining amyloids in its liquid interior, while they do not form outside the droplets. This was achieved by using a unique amphiphilic design which is part amyloid and part polyelectrolyte co-assembling with other liquid phase separating, charge complementary, polyelectrolytes. The design makes sure that the amyloids are tethered and confined to the liquid interior. This is of significant consequence as a minimalistic design feature for functional, multicomponent condensates that much like cellular environments take advantage of their heterogeneity to direct metabolic processes.
April 2022
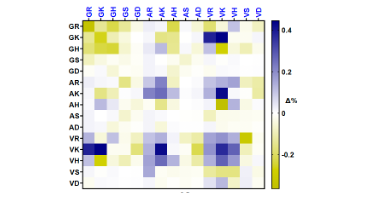
In this new paper, Ankit studied the collective properties of peptide mixtures composed of 10s to 100s of interacting peptides that can adapt their compositions to their environment. He developed a synthetic approach and analysis method to begin uncovering how complex biomolecule mixtures interact and collectively adapt to changes in their environment.
His experiment began with mixing a number of selected dipeptides, based on their ability to aggregate and interact. In the presence of thermolysin, a non-specific amidase known for its ability to hydrolyze and ligate dipeptides, the dipeptides dynamically recombine and form peptides with complex interaction patterns. It was then possible for Jain to track the formation and breakdown of peptides of different sequence within the mixtures. He observed that their patterns of interaction were strongly dictated by environmental conditions.
November 2021
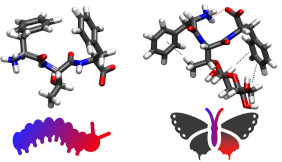
Glycosylation is a common biological post-synthetic modification significant, yet under-explored implications for enhancing functionality in designed supramolecular materials. In this JACS publication, we use simple self-assembling tripeptides containing polar amino acids Ser and Thr as sites for glycosylation. Striking observations from the computational modeling of these systems show that the conformational landscape of these simple tripeptides, typically pre-organized by F-F self-stacking interactions, is dramatically increased by glycosylation. This is underpinned by the generation of a diverse supramolecular interactome consisting of contributions from weaker interactions such as CH-π.
We also find striking and counter-intuitive differences between the two-polar amino acids studied, with the more hydrophobic Thr residue causing an overall reduction in aggregation propensity due to disruption of F/F hydrophobic collapse driven self-assembly by CH-π interactions. The glycosylation leads to increase in overall hydration of these peptides which is reflected in the changes in material properties such reduced formation of amyloid-like structures and enhanced thermostability. Therefore, we believe this to be a first step in understanding the changes in molecular interactions upon glycosylation and their implications in the design of self-assembling glycopeptide-based materials.
November 2021
Richard's latest paper demonstrates peptide-functionalized nanoparticles that can communicate with cancer cells and slow their development. The work - detailed in a newly published paper in Advanced Materials - is based charge-complementary zwitterionic peptides that give rise to remarkably robust self-assembly behavior in biological media.
The peptide design was incorporated into MMP-responsive peptides, so that nanoparticle self-assembly was activated when they encounter cancer cells. The cells take up the particle aggregates, and thereby instruct the cells to slow their growth. Because the nanoparticles communicate only with the cancer cells, healthy cells aren’t impacted.

May 2021
Rein has been awarded a U.S. Department of Defense's Vannevar Bush Faculty Fellowship - the agency's most prestigious single-investigator award. The five-year fellowship will provide $3 million to support Ulijn's work to understand how complex mixtures of molecules acquire functionality, and to repurpose this understanding to create new nanotechnology that is inspired by living systems.
Read more about the award.
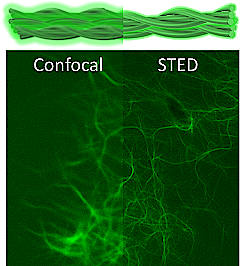
November 2020
As with most things in life, "seeing is believing" is also true in soft nanomaterials. Visualization of nanostructures with electron microscopy is limited to dry or frozen samples, whereas, traditional fluorescence microscopy has lower optical resolution and often involves a complex dye labeling procedure. The latest work by Mohit, with contributions from Jiye, Richard and Deborah demonstrates a new and remarkably simple method to fluorescently label peptide nanostructures for their visualization in solution with super-resolution microscopy. We show that by simply mixing a small amount (0.1 mole%) of a commercially available dye (Alexa) with cationic self-assembling peptides gives rise to selective electrostatic association of the dyes with the peptide nanostructure surface, which enables imaging using Stimulated Emission Depletion (STED) based super-resolution microscopy. Importantly, the method gave new insights into the hierarchical organization of peptide nanostructures, and enabled visualization of a metabolic transformation as demonstrated by enzymatic degradation of peptide nanofibers which could be imaged in real-time in situ. The ease and general scope of the method developed and the high resolution (approx. 50 nm) will open doors for potential in vivo imaging for bio-medical applications.

November 2020
We are interested in developing approaches to produce melanin-like materials with precisely controlled properties by using a bio-inspired approach, but radically simplified so that properties can be optimized specifically for technological applications, thus going beyond biologically available structures.
In this manuscript- by Eileen and Ayala in collaboration with the group of Nurit Ashkenasy at Ben Gurion University of the Negev, Israel, and Tell Tuttle and colleagues at the University of Strathclyde, UK, we demonstrate the use of a self-assembling tripeptide as a precursor for enzymatic formation of nanofibrous melanin-like structures with well-defined, and controllable electronic properties. We demonstrate two-fold conceptual novelty in (i) the ability to control oxidation but retain a well-defined fibrous morphology, that does not have a known equivalent in biology, and (ii) demonstrate unprecedented conductivity that is enhanced by enzymatic oxidation, and is demonstrated to be mediated by proton charge transfer.
September 2020
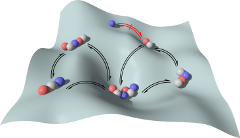
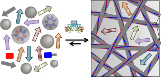
Water evaporation is a remarkably powerful process, providing a clean source of energy to power mechanical machines and devices. In a newly published paper in Nature Materials, PhD student Roxy Piotrowska and collaborators within CUNY and at The University of Strathclyde demonstrate the development of shape-shifting crystals that directly convert evaporation energy into powerful motions.
These water-responsive materials are based on supramolecular tripeptide crystals that contain nanoscale pores where water tightly binds, and these pores are interspersed with a molecular network of stiff and flexible regions that powerfully contract when humidity reaches a critical value. This results in the crystals temporarily losing their ordered patterns, until humidity is restored and the crystals regain their original shape. This newly designed process can be repeated over and over, and gives rise to a remarkably efficient method of harvesting evaporation energy to perform mechanical work.

April 2018
Mohit's latest paper in Nature Chemistry demonstrates the use of amino acids as chemical signals to enable the active editing of a self-assembling semiconducting structure, which results in temporal control over nanostructures and consequent transient electronic conductivity. The work will be of potential use in interfacing electronics with biological system.
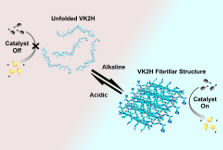
October 2017
Chunqiu's latest paper shows reversible regulation of catalytic activity of supramolecular peptide catalyst achieved by conformational transition from random coil to β-hairpin by changing the pH, which results in the reversible formation of an ordered array of 'active sites' composed of hydrophobic binding pockets and catalytic histidine residues with significant esterase activity. The work provides a step toward formation of self-regulating supramolecular assemblies.

June 2017
Melanins-the pigments that give color to skin, hair and eyes-have numerous useful qualities, including providing protection from cancer-causing UV radiation and free radicals, but also electronic conductance, adhesiveness and the capacity to store energy. We have developed a new approach for producing materials that not only mimic the properties of melanin, but also provide unprecedented control over expressing specific properties of the biopolymer, as published in Science. Unlike other biopolymers, such as DNA and proteins, where a direct link exists between the polymers ordered structures and their properties, melanin is inherently disordered, so directly relating structure to function is not possible. We found that the key to achieving polymers with controlled disorder is to start from systems that have variable order built in, which could be achieved by using self-assembling tripeptides with varying sequence as substrates for oxidative, biocatalytic polymerization. The discovery could enable the development of cosmetic and biomedical products.
October 2016
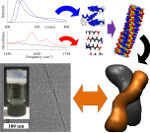
In this paper, published in Nature Nanotechnology, we demonstrate methodology to autonomously identify supramolecular polymers based on peptides. By allowing peptides to continuously reorganize their sequences, they will eventually form those polymers that are most suited to the environment at the expense of less favored structures. This methodology- dynamic peptide libraries, or DPL- is inspired by the principles of evolution, and allowed us to identify a range of heretofore unseen peptide-based materials with potential applications in drug delivery and cosmetics.
September 2016
In collaboration with the Tuttle group and recently published in Soft Matter, Ivan has combined computational and experimental methodologies to gain understanding on hierarchical self-assembly of Fmoc-dipeptides showing the lateral aggregation of fibers to form twisted ribbons and elucidating the interactions involved on this process. This study shows how MD simulations combined with spectroscopic and microscopy results enable development of atomistic models which then can be used to gain understanding of Fmoc-dipeptides nano structures.
August 2016
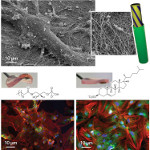
In this recent paper in new Cell Press journal Chem, which was the result of close collaboration with Matt Dalby, University of Glasgow, we describe the development of gels with tunable properties that mimic the nanofibrous structure and mechanical properties of biological tissue. These gels can be used to monitor stem cell differentiation and allow for the identification of molecules involved in differentiation.
June 2016
In Jugal's latest paper latest paper published in ACS Nano, in collaboration with Prof. Matthew J. Dalby's research group in University of Glasgow, we have used emerging rules of managing cell adhesion to prolong mesenchymal stem cell cultures by fabricating controllable nanoscale cell interfaces using immobilised peptides on a 2D surface, that may be enzymatically triggered to change their function. The surfaces can be altered at will to tip adhesion/cytoskeletal balance and initiate differentiation, hence better informing biological mechanisms of stem cell growth. This development of technology that is able to dynamically navigate small differences in cell adhesion is critical in the race to provide regenerative strategies using stem cells.
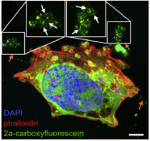
May 2016
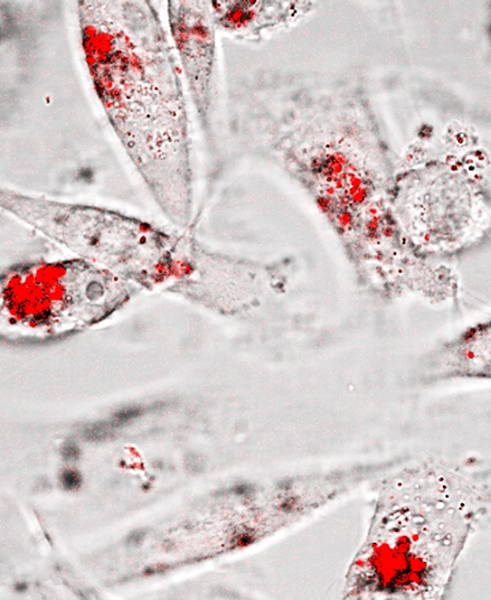
Daniela's latest paper in Biomaterials, in collaboration with the Beatson Cancer Research Institute (UK) shows peptide nanoparticles that change their shape when exposed to endogenous enzymes which are overexpressed in cancer. The morphology transition from spherical nano carrier to immobile nano fibers at the tumor site allows for localized delivery of anticancer drugs, which halts tumor growth. Thus, a pathological hallmark of metastasis (enzyme overexpression) can be exploited to guide therapeutic targeting, tuning a negative prognostic indicator into a therapeutic response.

February 2016
Front cover image of our recently discovered tripeptide emulsifiers. In this paper, we describe a new way to create emulsions with tunable properties, based on very simple biological molecules, as published in the journal Advanced Materials. In collaboration with Tell Tuttle's lab, we used a combination of experiments and simulations to show that dramatically variable oil-in-water emulsions may be produced from tripeptides.
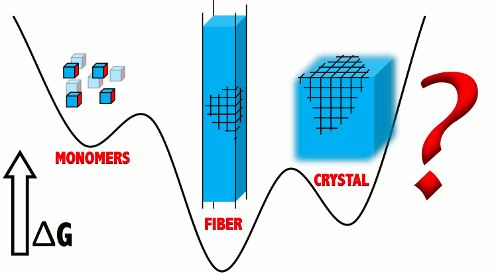
February 2016
In Ivan's latest paper latest paper, collaborating with the Tuttle group, published in ACS Nano we demonstrate that gels based in low molecular weight gelators (LWMG) can be at thermodynamic equilibrium, and hence, that not all of them are metastable. This study introduces a simple packing model which demonstrates that the amphiphilc nature of LMWGs is key to allow fiber networks to be more stable than crystals. This conclusion has critical implications for the theoretical treatment of gels.
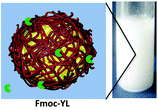
February 2016
Inês' latest paper shows on-demand formation of stable emulsions achieved by enzymatic activation. This is achieved using phosphate ester hydrolysis to trigger self-assembly, by conversion of a surfactant-type emulsion to a more permanent emulsion which is stabilized by a nanofibrous network at the oil-in-water droplet surface. The system is characterized using a combination of experimental and computational techniques.
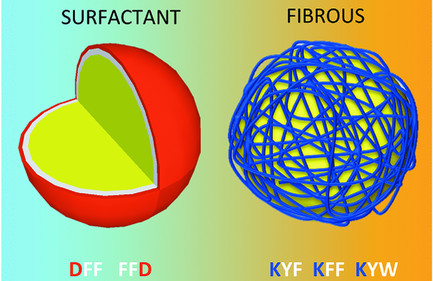
December 2015
In Gary's latest paper in collaboration with the Tuttle group we show a series of tripeptides that form emulsions with sequence tunable properties. Using a combination of simulations and experiments, it is shown that two types of oil-in-water emulsions may be produced, either forming stable interfacial nanofiber networks with remarkable stability, or more conventional surfactant-like monolayers. We are investigating applications in food, cosmetics and biomedicine.
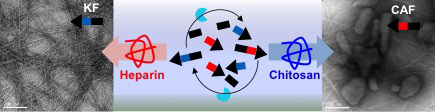
November 2015
In Yousef' latest paper published in Biomacromolecules, we show biomaterials that can dynamically adapt their structure and function by biocatalytic co-assembly. This is demonstrated by dynamic exchange of charged peptide sequences, to achieve selective amplification in the presence of cationic (chitosan) or anionic (heparin) biomacromolecules. The dynamic adaptive potential was demonstrated by sequential morphology changes depending on the sequence of polysaccharide addition. These results may pave the way to materials able to undergo dynamic morphogenesis.

November 2015
The Ulijn group features in the November edition of Bachem's Peptide Trends newsletter. The article profiles our work on biocatalytic peptides and designer peptide nanostructures.

August 2015
The Ulijn group's ASRC labs are quickly gathering critical mass. With the imaging suite and soft nano labs now up and running we are looking forward to a productive summer. Welcome to the new group members!

July 2015
Congratulations to David Velado on having his first year PhD work recognized by the American Chemical Society through a Certificate in Recognition for his "outstanding contributions to the future of chemistry". Nice job!

June 2015
The Ulijn group's ASRC labs are quickly gathering critical mass. With the imaging suite and soft nano labs now up and running we are looking forward to a productive summer. Welcome to the new group members!
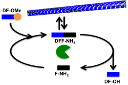
May 2015
In this paper published in Angewandte Chemie International Edition, we demonstrate sequence-dependent kinetic pathway selection in chemically fueled catalytic self-assembly of tripeptides. The systems are catalyzed by a-chymotrypsin and driven by hydrolysis of dipeptide aspartyl-phenylalanine-methyl ester (the sweetener aspartame, DF-OMe) in which the control of the lifetime of the nanostructures is dictated by chemical design. Mimicking the unique features of these systems may open up opportunities to create supramolecular systems for non-equilibrium motility and shape control.

December 2014
In this paper we use ultrasound, i.e. high frequency oscillating pressure waves, to achieve transient reorganization of supramolecular peptide nanostructures, which revert back to the original state when sound is switched off. The changes observed were due to an altered balance between H-bonding and π-stacking, giving rise in changes in chiral organisation of peptide building blocks.
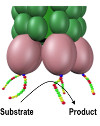
October 2014
In this paper, we introduce a new approach to catalyst discovery. This is achieved by combining catalytic self-assembly with phage display. We demonstrate the discovery of catalysts for amide condensation and hydrolysis in water, an extremely challenging reaction with an estimated uncatalyzed half-life in the range of 300 years. While oligopeptides have proven their worth as building blocks for molecular self-assembly and as selective ligands to achieve molecular recognition, our work provides a step towards applying them in design of minimalistic enzyme mimics.

October 2014
Joy's latest paper, published in ACS Nano, demonstrates nanopropulsion of enzyme-quantum dot conjugates by biocatalytic self-assembly. Specifically phosphatase enzymes act as the engine and quantum dots represent the vehicle. When exposed to micellar aggregates (fuel), biocatalytic dephosphorylation results in fibrillar self-assembly of aromatic dipeptide amphiphiles which in turn causes propulsion.

September 2014
After a decade of planning, the ASRC building has been completed. Today, the first researchers of the Nanoscience Initiative, under direction of Dr Rein Ulijn, have moved into the building.
The Nanoscience Initiative will occupy the ground and first floors. Research in Ulijn's group focuses on the development of biomimetic and adaptive nanoscale systems for biomedical and green energy applications. Over the next year they will be joined by further resident Faculty, affiliated members from across CUNY and a dynamic population of visiting scientists from the New York area and across the globe.
The Nanoscience Initiative aims to establish a world-leading research institute in computational and experimental nanoscience in Manhattan. This will be achieved through consolidating existing excellence in Nanoscience at the CUNY campuses coupled with major investment in new faculty, support staff, infrastructure and equipment. The $350-million Advanced Science Research Center (ASRC) is a nucleus for the nanoscience across The City University of New York, a diverse research community encompassing the five boroughs.
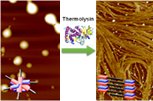
June 2014
In Krisha's latest paper published in Angewandte Chemie International Edition, we demonstrate the fabrication of 1D chiral charge-transfer nanofibers through the biocatalytic self-assembly of naphthalenediimide amino acid/dipeptide conjugate acceptors and dialkoxy/hydroxy naphthalene donors. In the obtained hydrogels, the charge-transfer-induced gel-sol-gel transformation led to major morphological changes. The dynamic nanostructures thus formed may provide opportunities for the development of aqueous adaptive nanotechnology.
Read more here.

March 2014
Rein Ulijn is moving to New York City in September 2014, joining the CUNY Advanced Science Research Center, now nearing completion. He will direct the Nanoscience Initiative which will focus on the development of biomimetic and adaptive nanosystems. Please contact Prof Ulijn for inquiries related to PhD, Postdoctoral and Faculty opportunities.
Read more here.

February 2014
Biogelx and Ulijn group research featured in Chemistry world. Rein and Elley explain the business and technology of cell matched gels that present well-defined chemistry in nanostructured gels. The gels help cell biologists address biological problems.
Read more here.

October 2013
Meghan's research article on the anti-microbial effects of morphologically different peptide nanostructures produced via intracellular biocatalytic self-assembly has been published on the front cover of Biomaterials Science.
Read the article here.

October 2013
Congratulations to Babis on his winning poster entitled 'Directing peptide self-assembly using sound waves', at the 1st Peptide Materials conference in Sorrento, Italy.

September 2013
Rein's 2013 RSC Norman Heatley Award has been profiled in Angewandte Chemie alongside other RSC award winners.
Read more here.
July 2013
In our new Chemical Science paper by Krishna, we report on the discovery of functional (rather than just structural) nanostructures using a dynamic combinatorial library (DCL) approach driven by an enzyme-assisted molecular self-assembly, demonstrating an enhanced self-selection and amplification of effective energy transfer nanostructures from complex mixtures of dipeptide derivatives. This approach enabled us to identify a two-component donor/acceptor hydrogel, which forms within minutes and exhibits efficient energy transfer.

July 2013
Our "science of jelly" outreach movie is now available online. A big thanks to all of the pupils and staff at the participating school and to Martin Laycock for filming and producing the movie.

June 2013

National Recognition for Biogelx!
Biogelx were among the winners of the first RSC Emerging Technologies Competition held at Burlington House on the 5th of June. The Award was presented to us by Universities and Science Minister David Willetts and RSC CEO Robert Parker.

April 2013
Rein wins 2013 RSC Norman Heatley Award!
The Norman Heatley Award recognises and promotes the importance of inter- and multi-disciplinary research between chemistry and the life sciences. It is named in honour of Norman Heatley who played a key role in the development of penicillin.
Professor Ulijn was awarded the prize in recognition of his pioneering work on the development of molecular materials that mimic aspects of biological systems, but are much simpler. He said: "This is wonderful recognition for my research group's hard work and dedication. This external recognition provides motivation and encouragement to continue our journey to develop new biologically inspired technologies and materials. Our objective is to translate some of these to real-world applications in nanotechnology and biomedicine over the next five years."

December 2012
biogelX Ltd formed!
The formation of biogelX Ltd., the commercial vehicle for Ulijn group gel technology was completed on the 18th of December.
More details on this exciting development will follow in the new year.

December 2012
EU Research Interview published!
Rein was recently interviewed by EU Research to discuss his ERC Starting grant on minimal biomimetic systems. An article based on this interview recently appeared in EU Research.
Find out more at the EU Researcher website or read the full article here.

November 2012
Strathclyde has been named University of the Year!
The University of Strathclyde has been named the UK's University of the Year in the 2012 Times Higher Education Awards. Judges described the University as a "bold, imaginative and innovative institution" and said the University's dedication to working with industry took concrete form in the past year, giving a modern slant to its 18th Century founder's vision of a 'place of useful learning'.
November 2012
Mischa's review article on enzyme-responsive materials has been published and made the front cover of the first issue of the new RSC journal Biomaterials Science!
Read more at the Biomaterials Science blog or see all the Ulijn group covers in the gallery on our publications page

November 2012
Members of the Ulijn group attended the Nanopeptide 2012 - Peptides as Nanomaterials & Biomaterials conference, held at the University of Manchester from the 12th to the 14th of November. The event, organised by Stephen Hoare, covered a wide range of topics involving peptide nanomaterials including, 'Peptide Design', 'Dynamic peptide systems and Self assembly' and 'Peptide/Polymer interactions'.

July 2012
On the 5th and 6th July the 5th Annual Ulijn Group 'Mega-Meeting' took place. Talks were given by each of the group members on a diverse range of topics from hydrogels for biomedical and green energy applications to the new catalytic peptides.

July 2012
Collaborative team is gelling well! A recent article reporting work from a three way collaboration between the groups of Neil Hunt, Rein Ulijn and Chris Pickett has been chosen as a Hot Article by the referees of the journal Dalton Transactions. The work reports the ultrafast dynamics of a model compound of the active site of the hydrogenase enzyme encapsulated within a peptide-based hydrogel. Such novel encapsulation confers not only significantly improved stability to the compound but also modified dynamical behaviour. Hydrogenase models are bio-inspired synthetic systems capable of catalysing the production of hydrogen and are seen as a new route to hydrogen fuel cell design. Read more here.

June 2012
Rein and Elley, together with collaborators Marie Boyd and Annette Sorensen won the 2012 Dragon's Den Event, part of Glasgow Science Week. The £20K award will be used to investigate the use of biomolecular gels to expand cancer stem cells.

May 2012
Another Marie Curie International Training Network grant on Supramolecular Gels has been awarded during which the Ulijn laboratory will be working with collaborators across Europe.
Two three year PhD research positions will be available from October 2012.

May 2012
The Ulijn group was awarded a new BBSRC grant to study Protein Stabilising Molecular Gels for Biosensing and Bioenergy Devices in collaboration with Prof Richard Cogdell, FRS (University of Glasgow) and Dr Neil Hunt (Physics).

April 2012
The Ulijn group enjoyed delicious food at the 3rd edition of the now traditional International Buffet. The venue was Prof. Ulijn's flat where the group was warmly welcomed. Once a year the group performs a large scale experiment and transforms the kitchen into a culinary laboratory where each member tries their skills at cooking.
Read more here.

March 2012
This March the Ulijn group enjoyed a whisky experience in the Auchentoshan distillery; the only distillery in Scotland to use triple distilling to produce their whisky. What better way to enjoy a relaxing afternoon together and get familiar with Scotland's culture then experience a distillery tour in the country of whisky?
Read more here.

January 2012
An International Training Network grant on Systems Chemistry has started during which the Ulijn laboratory will be working with collaborators across Europe.
Read more at the new website just launched here.

December 2011
The Ulijn group has been awarded a Bridging The Gaps (BTG) grant awarded for pilot study on the construction of reconfigurable soft matter for selective ion binding in collaboration with Neil Hunt, Maxim Federov and Charles Knapp.

December 2011
The Ulijn group has been awarded a £28K feasibility grant from the Korean Institute for Advancement of Technology award to facilitate new collaboration with Korean companies.

November 2011
Strathclyde University has approved the formation of BioGells, a start-up company which will aim to commercialise peptide gels for in vitro cell culture applications.

October 2011
Rein was awarded a $452k grant from US Air Force from the US Air Force Office of Scientific Research to investigate the electronic properties of self assembling peptide nanostructures.
September 2011
Meghan's paper on two-dimensional peptide nanostructures published in Soft Matter!
For more information about these nano-sheets access the full paper here.

March 2011
Congratulations to Lynsey who recently won two poster prizes at the 12th annual meeting of the Surface Science and Biologically Important Interfaces (SSBII) meeting held in Belfast and at the 6th annual meeting of the RSC Biomaterials Chemistry group held at Warwick University in January.
You can see Lynsey in front of her poster here.

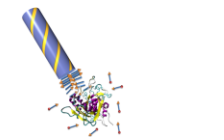
February 2011
Polymer Chemistry have identified a new paper from the Ulijn group as a Hot Article. The article describe a collaboration with the Univesity of Montreal to create polymer bioconjugates that are both thermally and enzymatically responsive. This article will feature in a forthcoming Emerging Investigators themed issue of Polymer Chemistry and is mentioned on their blog here.
October 2010
In a new Nature Chemistry paper, we show how we have been able to control the structural organization of self-assembling building blocks by enzymatic catalysis. A range of structures, representing local minima in the free energy landscape, could be accessed from a single precursor building block. Although it seems counter-intuitive, gels that assembled faster showed fewer defects.

September 2010
Rein has been awarded, with Brian Saunders from the University of Manchester, a £250K research grant from the EPSRC. The grant will fund two PhD students and will focus on the development of phosphatase responsive microgel systems for biomedical applications.

August 2010
Rein and Dek Woolfson are the co-editors of a new themed issue of Chemical Society Reviews: Peptide and protein based materials in 2010: from design and structure to function and application. This issue is packed with contributions from the leading laboratories in this exciting field.

August 2010
Rein Ulijn has been awarded a 1.5M Euro starting grant from the European Research Council. The grant will fund three postdoctoral researchers and will investigate "biocatalytic self-assembly" and its applications in biomedical and energy technologies.

July 2010
Lynsey's poster presentation was picked out as a highlight of the Challenges in Physical Chemistry and Nanoscience conference in Hungary by RSC blogger Philip Broadwith. He was impressed with her colorimetric elastase assay. Read more here.

March 2010
A new paper by Jan and Rein shows an enzyme-driven dynamic supramolecular peptide system which displays multiple reversible pathways. This system gives rise to emergent properties that are dictated by environmental conditions and that can be locked in a gel-state. Read more here.

March 2009
A new Ulijn group paper has been published in Small describing a method for controlling the solubility of carbon nanotubes by enzymes. Read more about this new use for Fmoc dipeptides here. The account is illustrated by an inside cover as seen here.

March 2009
A new Soft Matter paper has been published reporting on improved protease sensitive beads for enchanced response under physiological conditions. You can read the paper itself here, and Chemical Biology's comment here.

January 2009
Nature Nanotechnology paper published. You can find our paper on enzyme-catalysed reactions driving self-assembly here and a News & Views article by Sijbren Otto discussing the work here.
[Paper] [News & Views]

September 2008
Rein Ulijn appointed to new WestCHEM chair based at the University of Strathclyde in Glasgow.

July 2008
PSV Reindhoven triumph again in the MIB Football tournament! A reinforced Team Ulijn triumphed again despite Rein being sidelined by injury.

January 2008
Rein Ulijn wins Macro Group UK Young Researchers Medal, to be presented at the Frontiers of Research and Young Researchers Meeting at Warwick University
in April 2008.
[http://www2.warwick.ac.uk/fac/sci/chemistry/cim/research/dove/conferences/frontiers-yrm_flier.pdf]

September 2007
Read Rein Ulijn's Blog interview at Nature.com
[http://blogs.nature.com/thescepticalchymist/2007/09/reactions_rein_ulijn_1.html]

August 2007
Team Ulijn Triumph in inaugural MIB football tournament!

April 2007
Rein Ulijn receives Leverhulme Trust Research Leadership Award.
[http://www.mib.ac.uk/news.aspx]

February 2007
Rein Ulijn talks to Soft Matter about his latest paper "Enzyme-triggered cell
attachment to hydrogel surfaces":
[http://www.rsc.org/Publishing/Journals/sm/News/hot_ulijn.asp]

June 2006
Hydrogels research highlighted in Chemistry & Industry:
[http://www.chemind.org/CI/articles.jsp]

May 2006
Dr Ulijn’s research highlighted in Materials Today:
[http://www.materialstoday.com/pdfs/may_2006/research_news.pdf]
Enzyme-Responsive Materials research highlighted in Chemical Science:
[http://www.rsc.org/Publishing/ChemScience/Volume/2006/06/smart_materials.asp]

March 2006
Dr Ulijn interviewed by regenerationnet.com, the network for regenerative biology:
[http://www.regenerationnet.com/files/Rein.pdf]
[Older News]